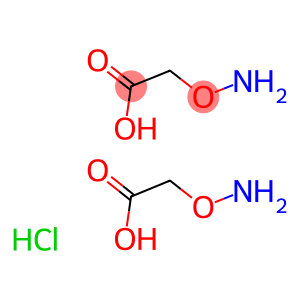
O-(Carboxymethyl)hydroxylamine hemihydrochloride,(Aminooxy)acetic acid hemihydrochloride, (Carboxymethoxy)amine hemihydrochloride, Hydroxylamine-O-acetic acid hemihydrochloride
Carboxymethoxylamine hemihydrochloride
CAS: 2921-14-4
Molecular Formula: C2H6ClNO3
O-(Carboxymethyl)hydroxylamine hemihydrochloride,(Aminooxy)acetic acid hemihydrochloride, (Carboxymethoxy)amine hemihydrochloride, Hydroxylamine-O-acetic acid hemihydrochloride - Names and Identifiers
O-(Carboxymethyl)hydroxylamine hemihydrochloride,(Aminooxy)acetic acid hemihydrochloride, (Carboxymethoxy)amine hemihydrochloride, Hydroxylamine-O-acetic acid hemihydrochloride - Physico-chemical Properties
| Molecular Formula | C2H6ClNO3 |
| Molar Mass | 127.53 |
| Density | 1.7848 (rough estimate) |
| Melting Point | 156°C (dec.)(lit.) |
| Boling Point | 326.7°C at 760 mmHg |
| Flash Point | 151.4°C |
| Water Solubility | decomposes |
| Vapor Presure | 4.24E-05mmHg at 25°C |
| Appearance | White crystal |
| Color | White to off-white |
| BRN | 3680528 |
| Storage Condition | 2-8°C |
| Sensitive | Easily absorbing moisture |
| Refractive Index | 1.6500 (estimate) |
| MDL | MFCD00012955 |
| Physical and Chemical Properties | White crystals. Melting point 156 °c (decomposition). |
| Use | It is used to introduce acidic groups into ketone compounds and can prepare chemiluminescent materials, such as preparing chemiluminescent substances ABEI labels on ketone compounds. It is also used as an inhibitor of transaminase. |
O-(Carboxymethyl)hydroxylamine hemihydrochloride,(Aminooxy)acetic acid hemihydrochloride, (Carboxymethoxy)amine hemihydrochloride, Hydroxylamine-O-acetic acid hemihydrochloride - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| RTECS | AF3150000 |
| FLUKA BRAND F CODES | 3 |
| HS Code | 29280000 |
O-(Carboxymethyl)hydroxylamine hemihydrochloride,(Aminooxy)acetic acid hemihydrochloride, (Carboxymethoxy)amine hemihydrochloride, Hydroxylamine-O-acetic acid hemihydrochloride - Reference
| Reference Show more | 1. Wang, Jieqi, et al. "Hydrophilic modification of PTFE microfiltration flat membrane by crosslinking OCMCS-PEI to enhance anti-fouling property." Progress in Organic Coatings 135 (2019): 565-573.https://doi.org/10.1016/j.porgcoat.2019.06.035 |
O-(Carboxymethyl)hydroxylamine hemihydrochloride,(Aminooxy)acetic acid hemihydrochloride, (Carboxymethoxy)amine hemihydrochloride, Hydroxylamine-O-acetic acid hemihydrochloride - Introduction
Decomposition in water, hygroscopic, can react with aldehydes to form fat.
Last Update:2022-10-16 17:24:18
O-(Carboxymethyl)hydroxylamine hemihydrochloride,(Aminooxy)acetic acid hemihydrochloride, (Carboxymethoxy)amine hemihydrochloride, Hydroxylamine-O-acetic acid hemihydrochloride - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| biological activity | AOA hemihydrochloride (Aminooxyacetic acid, Carboxymethoxylamine, AOAA) is an inhibitor of aminobutyric acid transaminase (aminobutyrate aminotransferase), which is involved in the metabolism of amino acids and polyamines. |
| Target | Value |
| use | organic synthesis intermediate. It is used to introduce acidic groups into ketone compounds and can prepare chemiluminescent materials, such as preparing chemiluminescent substances ABEI labels on ketone compounds. It is also used as an inhibitor of transaminase. |
| Production method | O-carboxymethylacetone oxime is obtained by reacting with hydrochloric acid. |
Last Update:2024-04-09 02:00:08
Supplier List
Spot supply
Product Name: Carboxymethoxylamine hemihydrochloride Visit Supplier Webpage Request for quotationCAS: 2921-14-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: O-(Carboxymethyl)hydroxylamine hemihydrochloride Visit Supplier Webpage Request for quotationCAS: 2921-14-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Carboxymethoxylamine hemihydrochloride Request for quotation
CAS: 2921-14-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2921-14-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: O-(Carboxymethyl)hydroxylamine hemihydrochloride Visit Supplier Webpage Request for quotationCAS: 2921-14-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Carboxymethoxylamine hemihydrochloride Visit Supplier Webpage Request for quotationCAS: 2921-14-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: O-(Carboxymethyl)hydroxylamine hemihydrochloride Visit Supplier Webpage Request for quotationCAS: 2921-14-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Carboxymethoxylamine hemihydrochloride Request for quotation
CAS: 2921-14-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2921-14-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: O-(Carboxymethyl)hydroxylamine hemihydrochloride Visit Supplier Webpage Request for quotationCAS: 2921-14-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



